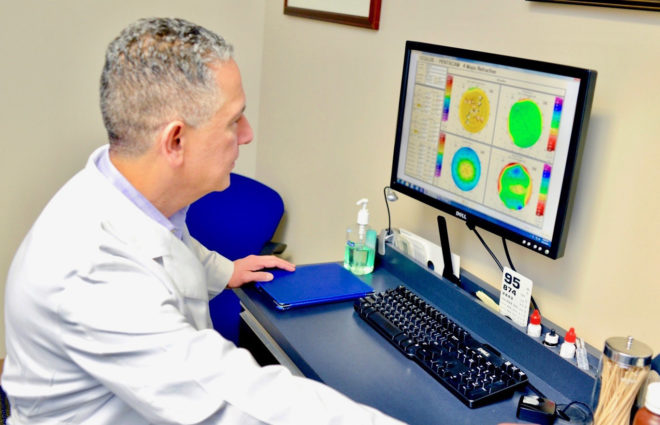
Dr. Mozayeni, Cornea Specialist, Clinical trial physician
At Providence Eye & Laser Specialists, we are deeply committed to the advanced treatment of keratoconus and providing FDA-approved options as they become available. We are honored to have been one of the more than 20 sites across the United States participating in the Phase 3 FDA clinical trial of Epi-ON corneal cross-linking, known as EpiSmart™. While enrollment for this study at our practice is now complete, we are excited to watch the progress of EpiSmart over the next few years.
Keratoconus (kehr-uh-toh-koh-nus) is a condition where the cornea—the clear, dome-shaped surface of the eye—thins and becomes irregularly shaped, causing a cone-like bulge. This results in distorted vision and symptoms such as:

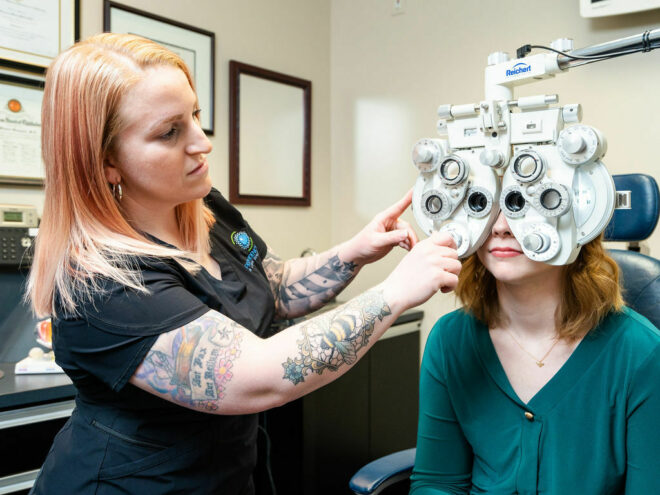
Epi-ON cross-linking is a less invasive procedure that strengthens the cornea, slowing or halting the progression of keratoconus. This method leaves the cornea’s outer layer intact, offering several benefits over the traditional Epi-OFF approach:
Dr. Reza Mozayeni, Dr. Trey Nunnery and the team at Providence Eye & Laser Specialists are dedicated to helping patients with keratoconus through innovative treatments like Epi-ON corneal cross-linking.
Although our participation in the EpiSmart clinical trial is complete, Providence Eye remains committed to following developments in keratoconus treatment to bring the latest advancements to our patients.
Call (704) 540-9595 or fill out this short form to be contacted by a member of our team.